Geminal Theory
Modern electronic structure methods can be divided into two groups: simple single-reference
methods (Hartree-Fock, Coupled Cluster, Kohn-Sham Density Functional Theory) and "custom-made"
multireference methods (MCSCF, CASPT2). Single-reference methods work well for equilibrium
ground states of not very reactive species, and multireference methods are often computationally
expensive and are hard for non-theorists to use. Our group develops a well-defined
method based on geminal representation of the wave function. Its full name is Antisymmetrized Product of
Singlet type Strongly orthogonal Geminals (APSSG), and SSG is the shorter acronym.
The SSG method is the only practical model that is both variational and size consistent.
Its computational cost is only modestly higher than the versatile Hartree-Fock. The
target applications of SSG are chemistry of transition metal elements and potential
energy surfaces (reaction barriers, bond energies). SSG model is implemented in Q-Chem, and the geminals can be visualized with IQMol (Fig1).
The factorized electron-nuclear quantum dynamics
The nuclear quantum effects are commonly described in chemical applications through
the transitions between the Born-Oppenhemer (adiabatic) states. This approach becomes
cumbersome when the number of electronic states involved in the process is large.
The alternative approach explored by the number of research groups around the world
is based on a single time-dependent electronic state governing the molecular dynamics
(Fig 2).
Computational chemistry
Computational tools have become an indispensable part of much of chemical research.
Yet the answers provided by the computations can be misleading, or sometimes just
plain wrong. Successful computational research relies on prior experience (community
or in-house), and the close interaction with experiment. The computational research
in our group is in the areas of, an in collaboration with:
- Mechanisms of C-H bond activation by metal carbonyl complexes (Adams)
- Dynamics of excited states in metal organic frameworks (Shustova)
- Stability of substituted metallocenium compounds (Tang) Fig. 3
- Rotational energy barriers in small organic molecules (K. Shimizu)
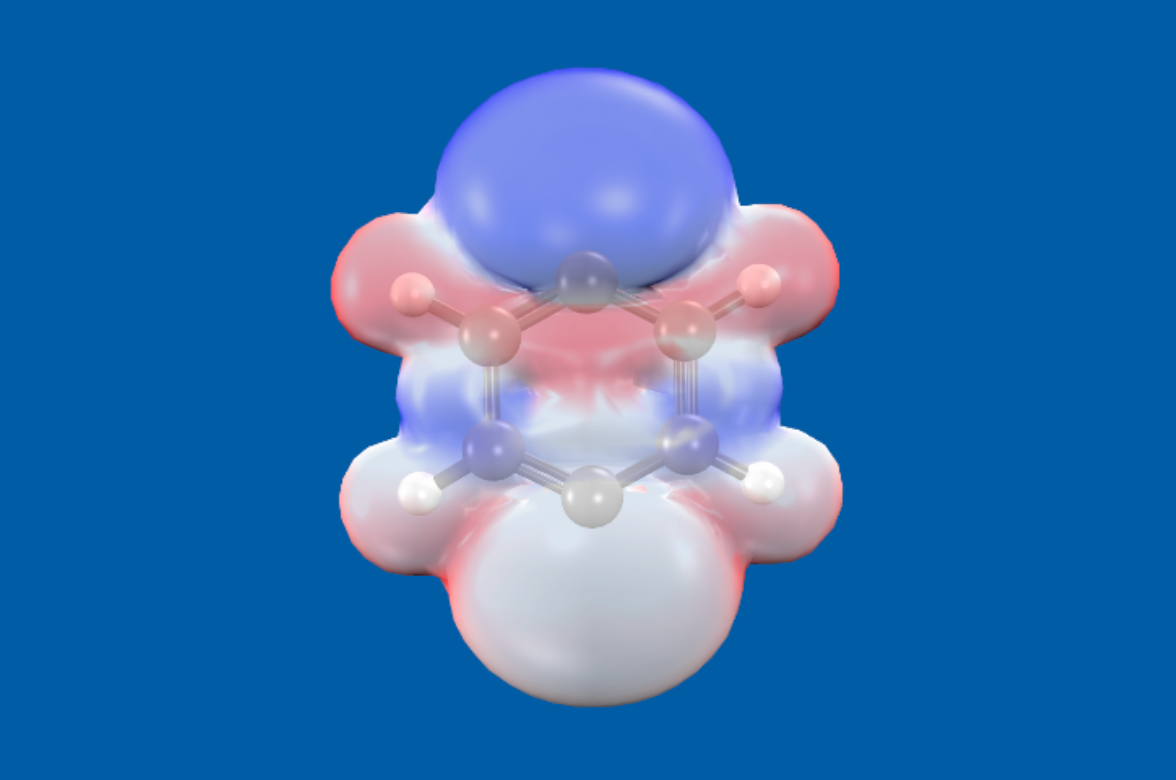
Fig.1

Fig.2

Fig.3